As you will know, the current 9-1 GCSEs examine practical work through exam questions. The questions cover different aspects of the specification and particular apparatus and techniques, and these are listed alongside each of the practicals detailed in each exam specification handbook.
It is important to remember that practicals are a vehicle for teaching about the apparatus and the techniques specified, and that it is the minimum that students should be exposed to.
Be clear on purpose
Before carrying out a practical, it helps to be sure why we are carrying it out.
-
Do we want students to become proficient at handling a piece of equipment (for example, a burette or pipette)?
-
Do we want students to get a set of results to use (for example, finding the distribution of organisms in an area)?
-
Or do we want to evaluate a particular method (for example, by finding the specific heat capacity of a metal block and comparing the value obtained experimentally with published data)?
By using the published technician results when modelling calculations, you can ensure that all students should get the same answers, thereby simplifying the process of instruction. Students can then be given follow-up calculations to do to build their proficiency and to give them a chance to select the correct results to include how to calculate means, rearrange equations and convert units.
Reducing cognitive load
Much has been written about cognitive load (the way that information is presented to a student) and it can be helpful to reduce this by simplifying instructions. An easy way to do this is to provide practical instructions as a set of diagrams (with measurements such as volume of solution required). Students can then carry out the practical and can also use the visual prompts to help them recall and record a method. Diagrammatic cards can also be adapted to create a card sort to sequence the stages of the reaction – a useful aid for revision purposes.
Selecting equipment
Working scientifically skill 2.3 (detailed in your exam specification) requires the selection of apparatus or techniques to carry out a particular practical.
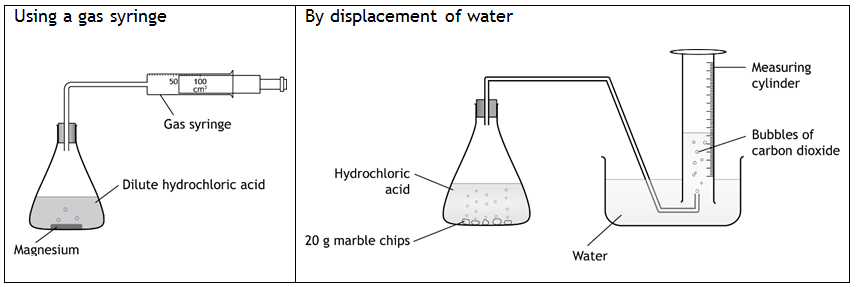
A simple way of building this skill with students is to provide a range of different pieces of equipment for some practicals – for example, a range of measuring cylinders and beakers of different volumes to measure a given volume of solution. Alternatively, show students how to carry out a practical using different apparatus – measure the volume of gas produced by displacement of water or in a gas syringe, for example, or the practical could be set up to show the loss of mass as the gas escapes from the reaction vessel, by placing the un-stoppered reaction vessel on a balance and recording the mass at regular intervals.
Application
Another feature of the new GCSE practicals is the increase in weighting of assessment objective 2 (AO2), the application of knowledge and understanding. When planning a practical, it is worth considering how to teach AO2. For example, when making a salt from an insoluble oxide or carbonate, consider giving different students different combinations. For instance, give some students copper oxide and hydrochloric acid, and give others copper carbonate and sulphuric acid.
Then ask them to predict the salts that would be made using other combinations of acids and bases. The process remains the same – warm the acid, react the acid and base, filter to remove the unreacted base and then heat over a water bath, and finally leave to cool and crystallise.
Evaluation
Evaluation is a higher-order skill and one that many students struggle with. Try to provide opportunities for students to consider ways to improve experimental technique. Start with the more straightforward practicals – such as those which involve heat loss to the surroundings (exothermic reactions, measuring specific heat capacity, combustion of fuels, thermal insulation, and so on) and use simple equipment with minimal insulation. Provide a range of alternatives such as polystyrene cups, lids, and so on, and encourage students to consider the impact of using these. Try and demonstrate the use of digital equipment such as thermometers or temperature probes, which can read to the nearest decimal place and not just 0.5°C.
The language of science
Ensure students are clear about the meanings of words and be pedantic about their use of the correct terms. Make card sorts or other activities where students have to match the term to the correct definition and consider building some working scientifically slides into lessons.